-
HOW LIQUIDS CONDUCT ELECTRICITY? –ACTIVITY
HOW LIQUIDS CONDUCT ELECTRICITY? –ACTIVITY
To test whether a liquid allows electric current to pass through it or not, we can use the same tester (Fig.14.1).
Figure 14.1: a tester-good conductor
Figure 14.1: a tester-bad conductor
However, replace the cell by a battery. Also, before using the tester we should check whether it is working or not.
Activity 14.1:
* This completes the circuit of the tester and the bulb should glow.
* However, if the bulb does not glow, it means that the tester is not working.
* Can you think of the possible reasons? Is it possible that the connections are loose?
* Or, the bulb is fused? Or, your cells are used up? Check that all the connections are tight.
* If they are, then replace the bulb with another bulb.
* Now test if the tester is working or not.
* If it is still not working then replace the cells with fresh cells.
Now that our tester is working, let us use it to test the various liquids.
(Caution: While checking your tester, do not join its free ends for more than a few seconds. Otherwise, the cells of the battery will drain very quickly.)
Activity 14.2: ( Conduction in lemon juice )
* Collect a few small plastics or rubber caps of discarded bottles and clean them.
* Pour one teaspoon of lemon juice or vinegar in one cap.
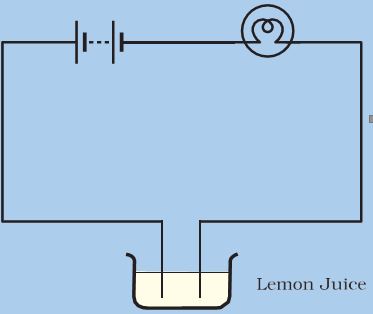
* Bring your tester over this cap and let the ends of the tester dip into lemon juice or vinegar as shown in Fig.14.2.
* Take care that the ends are not more than 1 cm apart but at the same time do not touch each other.
* Does the bulb of the tester glow? Does lemon juice or vinegar conduct electricity?
* How would you classify lemon juice or vinegar— a good conductor or a poor conductor?
Figure 14.2: Testing conduction of electricity in lemon juice or vinegar
When the liquid between the two ends of the tester allows the electric current to pass, the circuit of the tester becomes complete. The current flows in the circuit and the bulb glows. When the liquid does not allow the electric current to pass, the circuit of the tester is not complete and the bulb does not glow.
In some situations even though the liquid is conducting, the bulb may not glow. It may have happened in Activity14.2. What can be the reason?
Do you remember why the bulb glows when the electric current passes through it? Due to the heating effect of the current, the filament of the bulb gets heated to a high temperature and it starts glowing. However, if the current through a circuit is too weak, the filament does not get heated sufficiently and it does not glow. And why is the current in the circuit weak? Well, though material may conduct electricity, it may not conduct it as easily as a metal. As a result, the circuit of the tester may be complete and yet the current through it may be too weak to make the bulb glow. Can we make another tester which can detect a weak current?
We can use another effect of an electric current to make another kind of tester. Do you recall that electric current produces a magnetic effect? What happens to a compass needle kept nearby when current flows in a wire? Even if the current is small, the deflection of the magnetic needle can be seen. Can we make a tester using the magnetic effect of currents? Let us find out in Activity 14.3.
Activity 14.3:
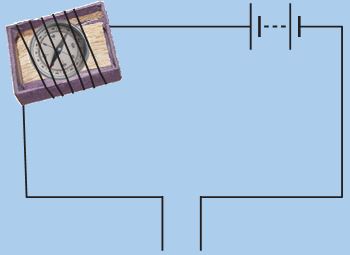
* Take the tray from inside a discarded matchbox.
* Wrap an electric wire a few times around the tray.
* Place a small compass needle inside it.
* Now connect one free end of the wire to the terminal of a battery.
* Leave the other end free.
* Take another piece of wire and connect it to the other terminal of the battery (Fig. 14.4).
Figure 14.4: Another tester
* Join the free ends of two wires momentarily.
* The compass needle should show deflection.
* Your tester with two free ends of the wire is ready.
* Now repeat Activity 14.2 using this tester.
* Do you find a deflection in the compass needle the moment you dip the free ends of the tester in lemon juice?
* Take out the ends of the tester from the lemon juice, dip them in water and then wipe them dry.
* Repeat the activity with other liquids such as tap water, vegetable oil, milk, honey.
(Remember to wash and wipe dry the ends of tester after testing each liquid).
* In each case observe whether the magnetic needle shows deflection or not.
* Record your observations in Table 14.1.
S.No
Material
Compass Needle
Shows Deflection
Yes/No
Good Conductor/
Poor Conductor
1.
Lemon juice
Yes
Good Conductor
2.
Vinegar
3.
Tap Water
4.
Vegetable oil
5.
Milk
6.
Honey
7.
8.
9.
10.
Table 14.1: Good-Poor Conducting Liquids
From Table 14.1, we find that some liquids are good conductors of electricity and some are poor conductors.
When the free ends of the tester do not touch each other, there is an air gap between them. Paheli knows that air is a poor conductor of electricity. But she has also read that during lightning, an electric current passes through the air. She wonders if the air is indeed a poor conductor under all conditions. This makes Boojho ask whether other materials classified as poor conductors also allow electricity to pass under certain conditions.
Actually, under certain conditions, most materials can conduct. That is why it is preferable to classify materials as good conductors and poor conductors instead of classifying as conductors and insulators.
We have tested the conduction of electricity through tap water. Let us now test the conduction of electricity through distilled water.
Activity 14.4: ( distilled water-salt )
* Take about two teaspoonfuls of distilled water in a clean and dry plastic or rubber cap of a bottle.
(You may obtain distilled water from your school science lab. You may also get distilled water from a medical store or a doctor or a nurse).
* Use the tester to test whether distilled water conducts electricity or not.
* What do you find? Does distilled water conduct electricity?
* Now dissolve a pinch of common salt in distilled water.
* Again test. What do you conclude this time?
When salt is dissolved in distilled water, we obtain salt solution. This is a conductor of electricity.
The water that we get from sources such as taps, hand pumps, wells and ponds is not pure. It may contain several salts dissolved in it. Small amounts of mineral salts are naturally present in it. This water is thus a good conductor of electricity. On the other hand, distilled water is free of salts and is a poor conductor.
Small amounts of mineral salts present naturally in water are beneficial for human health. However, these salts make water a good conductor. So, we should never handle electrical appliances with wet hands or while standing on a wet floor.
We have found that common salt, when dissolved in distilled water, makes it a good conductor. What are the other substances which, when dissolved in distilled water, make it conducting? Let us find out.
Caution: Do the next activity under the supervision of your teacher/parent or some elderly person, because the use of acid is involved in it.
Activity 14.5: ( distilled water-acid-base-sugar )
* Take three clean plastic or rubber caps of bottles.
* Pour about two teaspoonfuls of distilled water in each of them.
* Add a few drops of lemon juice or dilute hydrochloric acid to distilled water in one cap.
* Now in the second cap containing distilled water, add a few drops of a base such as caustic soda or potassium iodide.
* Add a little sugar to the distilled water in the third cap and dissolve it.
* Test which solutions conduct electricity and which do not.
* What results do you obtain?
Most liquids that conduct electricity are solutions of acids, bases, and salts.
When an electric current flows through a conducting solution, does it produce an effect on the solution?
You may use an LED (Fig. 14.3) in place of the electric bulb in the tester of Fig. 14.2. LED glows even when a weak electric current flows through it.
There are two wires (called leads) attached to an LED. One lead is slightly longer than the other. Remember that while connecting to a circuit, the longer lead is always connected to the positive terminal of the battery and the shorter lead is connected to the negative terminal of the battery.
Figure 14.3: LEDs
Source: This topic is taken from NCERT TEXTBOOK